Heisenberg principle of observation12/27/2023   
In short: the uncertainty principle describes a trade-off between two complementary properties, such as speed and position. Conversely, if we wanted to know the exact position of one peak of a wave, we would have to monitor just one small section of the wave and would lose information about its speed. Simply let p mv, and x h / (mv) (from De Broglie’s expression for the wavelength of a particle). Equation 1.9.4 can be derived by assuming the particle of interest is behaving as a particle, and not as a wave. 
The location is spread out among the peaks and troughs. or, px hearly form of uncertainty principle. The observer effect simply states that we change the. The more peaks and troughs that pass by, the more accurately we would know the speed of a wave-but the less we would be able to say about its position. From the Heisenberg Principle we drifted into the Copenhagen interpretation of Quantum Physics and (last but not least) the Observer Effect. To measure its speed, we would monitor the passage of multiple peaks and troughs. To understand the general idea behind the uncertainty principle, think of a ripple in a pond. Quantum objects are special because they all exhibit wave-like properties by the very nature of quantum theory. His most well-known thought experiment involved photographing an electron. 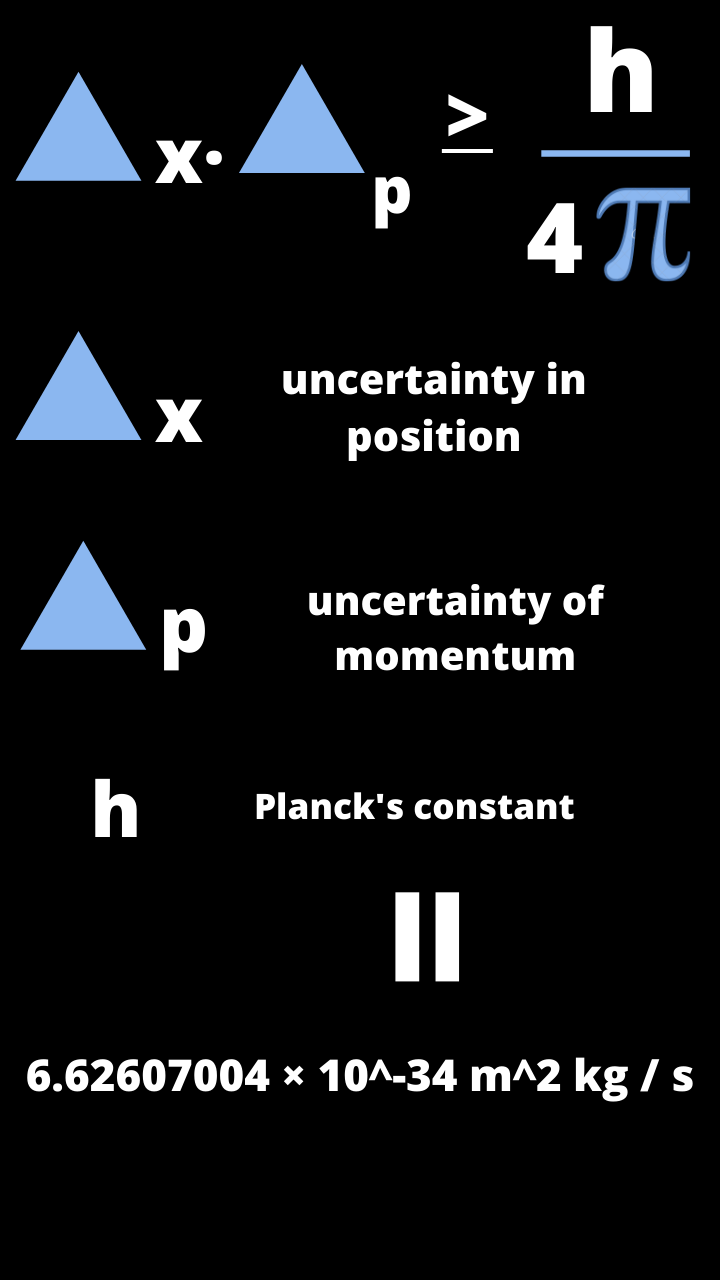
Though the Heisenberg uncertainty principle is famously known in quantum physics, a similar uncertainty principle also applies to problems in pure math and classical physics-basically, any object with wave-like properties will be affected by this principle. Heisenberg sometimes explained the uncertainty principle as a problem of making measurements. In other words, if we could shrink a tortoise down to the size of an electron, we would only be able to precisely calculate its speed or its location, not both at the same time. Of these scales, the most we can hope for is to calculate probabilities for where things are and how they will behave.Formulated by the German physicist and Nobel laureate Werner Heisenberg in 1927, the uncertainty principle states that we cannot know both the position and speed of a particle, such as a photon or electron, with perfect accuracy the more we nail down the particle's position, the less we know about its speed and vice versa. The uncertainty principle says there is a fundamental limit to what we can know about the behaviour of quantum particles and, therefore, the smallest scales of nature. Singh said that Capra has mentioned that Heisenberg got the fundamentals of his principle while in a conversation over Vedas with Rabindranath Tagore. The press note read that Singh said, "Famous scientist Eisenhower's theory of uncertainty was based on Vedas." However, a careful listening of the minister's speech shows that he actually mentioned "Heisenberg" and not "Eisenhower". But the home ministry's press note on the issue made the minister look rather ignorant. The statement, made on Hindi Diwas, is based on assertions made in a 1975 book by Fritjof Capra. Formulated by the German physicist and Nobel laureate Werner Heisenberg in 1927, the uncertainty principle states that we cannot know both the position and speed of a particle, such as a photon or electron, with perfect accuracy the more we nail down the particle's position, the less we know about its speed and vice. Heisenbergs uncertainty principle is a very precise mathematical statement about the nature of a quantum system. NEW DELHI: In line with BJP's consistent emphasis on projecting the country's ancient knowledge, home minister Rajnath Singh on Saturday said that fundamentals of German physicist Werner Heisenberg's principle of uncertainty (in quantum physics) were based on Vedas. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
